Alessandro Brunelli; Michele Salati
Curr Opin Pulm Med. 2008;14(4):275-281. ©2008 Lippincott Williams & Wilkins
Posted 12/12/2008
The objective of this review is to summarize the best clinical evidence published in the last year and regarding the functional evaluation and the residual quality of life (QOL) after lung resection in patients with lung cancer. A recent evidence-based guidelines paper from the American College of Chest Physicians (ACCP)[1] highlights the importance of respiratory and ergometric tests in predicting the occurrence of postoperative complications and residual function. Most recent published evidences are presented herein and add to the existing guidelines.
Current Algorithms
The second edition of the ACCP functional evaluation guidelines in lung-resection candidates[1] recommend that for those patients with a preoperative forced expiratory volume in 1 s (FEV1) less than 1.5-2 l or 80% of predicted, predicted postoperative lung function should be calculated (by lung perfusion scan for pneumonectomy candidates and by anatomical segmental method for all the other lung resections). Patients with predicted postoperative forced expiratory volume in 1 s (ppoFEV1) less than 30% or a predicted postoperative product [ppoFEV1 x predicted postoperative carbon monoxide lung diffusion capacity (ppoDLCO)] less than 1650 are deemed inoperable.
In cases of patients with ppoFEV1 or ppoDLCO less than 40%, a cardiopulmonary exercise test (CPET) is recommended. If the VO2max is less than 10 ml/kg/min or in cases of VO2max lower than 15 ml/kg/min associated with a ppoFEV1 and ppoDLCO less than 40%, the surgical risk is regarded as prohibitive. In cases of a ppoFEV1 less than 30%, patients are deemed inoperable irrespective of any other tests.
This revised algorithm confirms the central role of ppoFEV1 for selecting patients for operation and for deciding additional tests.
Limitations of Predicted Postoperative Forced Expiratory Volume in 1 S
A recent paper from Brunelli et al.[2] showed that ppoFEV1 was a significant predictor of cardiopulmonary complications only in patients without airflow limitations (FEV1 > 70%) submitted to major lung resection but not in those with FEV1 less than 70%. These findings may be partly explained by the so-called ‘lung volume reduction effect’. In lung cancer patients with moderate to severe chronic obstructive pulmonary disease (COPD), the resection of the most affected parenchyma may determine an actual improvement in the elastic recoil, a reduction of the airflow resistance, and an improvement in pulmonary mechanics and V/Q matching. In this regard, many studies have already shown the minimal loss or even the improvement in pulmonary function after lobectomy in these obstructed patients, questioning the traditional operability criteria mostly based on pulmonary parameters.[3,4]
Most recently, Brunelli et al.[4] showed that COPD patients [FEV1 < 80% + FEV1/forced vital capacity (FVC) < 0.7] had significantly lower percentage losses of FEV1 and DLCO compared with non-COPD patients 3 months after lobectomy (8 versus 16% and 3 versus 12%, respectively). Furthermore, their exercise tolerance evaluated by stair-climbing test was similar to the one observed in non-COPD patients with a calculated VO2peak which returned to baseline preoperative values. In this series, 27% of COPD patients actually improved their FEV1, 34% their DLCO, and 43% their VO2max 3 months after operation.
In another paper, Brunelli et al.[5] found that the actual to predicted postoperative (apo/ppo) FEV1 ratio in COPD patients was 1.15, 3 months after lobectomy. They also found that not only FEV1 but also DLCO was better than predicted after lobectomy in COPD patients. In fact, the apo/ppo DLCO ratio was 1.22, 3 months after lobectomy.
Brunelli et al.[6] demonstrated that the lung volume reduction effect takes place very early after lung resection. In fact, in a group of 161 patients evaluated at discharge (median 8 days) after lobectomy, they found that in patients with a preoperative FEV1 less than 70% the mean FEV1 loss was only 12.6% versus a mean loss of 30% in those with FEV1 more than 70%. Seventeen percent of the obstructed patients actually improved their FEV1 at discharge.
This finding was subsequently confirmed by Varela et al.,[7] who showed that the percentage loss of FEV1 on the first postoperative day after lobectomy was lower in patients with lower COPD index, a parameter inversely correlated with the degree and purity of obstructive disease.
These findings show that ppoFEV1 may not work properly in those patients with obstructive disease, and cannot be used alone to select patients for operation, especially those with limited pulmonary functions.
Although ppoFEV1 has been shown to correlate fairly well with the definitive residual FEV1 at 3-6 months after the operation, it has been recently demonstrated that it substantially overestimates the actual FEV1 observed in the first postoperative days, when most of the complications occur.[8]
A prospective consecutive series of 125 uneventful lobectomy patients were analyzed by serial measurements of FEV1 preoperatively and during the 6 postoperative days until discharge by a portable spirometer. The actual FEV1 on the first postoperative day was about 30% lower than predicted. This may have serious clinical implications whenever using ppoFEV1 for patient selection and risk stratification before operation.
Varela et al.[9] demonstrated through a classification tree analysis (CART) that the FEV1 measured on the first postoperative day was a better predictor of complications compared with ppoFEV1 (100 versus 43% importance as primary splitter or surrogate of other variables, respectively).
With the aim to improve the accuracy of risk stratification, Brunelli et al.[10] developed and validated a model to estimate the value of FEV1 on the first postoperative day after major lung resection. In an external validation set, the mean predicted first-day postoperative FEV1 value did not differ from the observed one (42.6 versus 42.0, respectively; P = 0.3). If future analyses will prove its role in stratifying the early postoperative risk, this newly developed model may be integrated in preoperative evaluation algorithms to refine risk stratification.
The Role of Carbon Monoxide Lung Diffusion Capacity
Ferguson et al.[11] were the first to demonstrate DLCO to be an independent predictor of postoperative mortality and pulmonary morbidity. They identified increased mortality in patients with a DLCO of less than 60% of normal, as well as a twofold to threefold increased risk of pulmonary morbidity if the DLCO was less than 80%. In parallel to FEV1, also for DLCO, a predicted postoperative value has been proposed as possible predictor of complications. A cut-off of ppoDLCO less than 40% has been associated with increased surgical risk.[12,13]
One issue that is controversial is whether DLCO should be measured routinely, or only in patients who have compromised spirometric function. FEV1 and DLCO are thought to be complimentary pulmonary function tests, given that each assesses a distinct physiologic function of the lungs: airflow versus gas exchange. FEV1 is still considered by many surgeons to be the definitive pulmonary function test to perform prior to lung resection, and many clinicians will not perform DLCO measurement on patients whose FEV1 is greater that 80% of predicted normal. In the Thoracic Database of the Society of Thoracic Surgeons, only 57% of patients undergoing major lung resection from 2000 to 2006 had DLCO values reported.[14] More striking was the finding from the European Thoracic Surgery Database that only 23% of surgeons reported DLCO values for their major lung-resection patients.[15] Although, recent guidelines support this attitude,[1,16] more recent data show that DLCO is very important in predicting postoperative complications, even in patients without impaired FEV1 (<80% of predicted) or chronic obstructive lung disease (as defined by an FEV1/FVC ratio <0.7).[13,17] In fact, DLCO is an equally strong predictor of postoperative complications in patients with and without COPD. These findings indicate that DLCO should be routinely measured during preoperative evaluation regardless of whether or not the spirometric evaluation is abnormal.
Exercise Tests
Exercise testing is increasingly used to assess the aerobic reserve of lung-resection candidates. In fact, these tests have the capability to assess the oxygen transport system in its wholeness and to detect possible deficits that may predispose to postoperative complications.
Several forms of exercise are currently used in clinical practice for preoperative risk stratification.
Simple low-technology exercises are the shuttle-walk test and the stair-climbing test.
The shuttle-walk test appears more reproducible than the 6-min walk test. It has been estimated by regression analysis that 25 shuttles on the shuttle-walk test indicate a VO2peak of 10 ml/kg/min[18] and therefore this cut-off value has been suggested in the functional algorithm proposed by the British Thoracic Society (BTS)[19] as selection criterion for surgery.
However, recently Win et al.[20] found no statistically significant difference in shuttle-walk distance between patients with and without complications after lung resection and that it tends to underestimate exercise capacity at the lower range compared with peak oxygen consumption.[21] They concluded that this test should not be used to predict poor surgical outcome in lung cancer patients contrary to current recommendations from BTS.[19]
Stair-climbing test has been recently found to be a reliable predictor of postoperative complications and mortality following lung resection.[22,23]
In particular, in a series of 160 patients, we[23] found that the altitude reached at stair-climbing test was the only independent predictor of cardiopulmonary complications after controlling the effect of other variables by logistic regression analysis.
More recently, in a series of 640 patients submitted to major lung resection, we found that patients climbing less than 12 m had morbidity, mortality and costs twofold, 13-fold and 2.5-fold higher than patients climbing more than 22 m.[24] The mortality of patients climbing more than 22 m was only 1%. Even in patients with prohibitive pulmonary functions (ppoFEV1 or ppoDLCO or both less than 40%) the mortality in patients climbing more than 22 m was 0, versus a mortality of 20% in those not able to reach 12 m.
In addition to the altitude reached at stair-climbing test, we recently found that the presence of an exercise oxygen desaturation (EOD) greater than 4% was a strong predictor of adverse outcome after adjusting its effect for other factors with regression analysis. The mortality in patients experiencing EOD greater than 4% was 8% (versus 3% in those not experiencing it). Those patients who are able to climb less than 12 m with EOD greater than 4% had complication and mortality rates of 71 and 29%, respectively.[25]
Recently, some authors demonstrated a strong correlation between the speed of ascent during stair climbing and the actual VO2max measured during cycle ergometry. All patients with a speed greater than 15 m/min had a VO2max greater than 20 ml/kg/min. On the contrary, 98% of patients with a speed less than 12 m/min had a VO2max less than 15 ml/kg/min.[26]
High-technology exercise tests have several advantages over the much simpler ‘low-technology’ exercise tests. These tests are performed in a controlled environment with continuous monitoring of various parameters. This ensures easy standardization and good reproducibility of results. In addition to VO2max, a number of other cardiac and respiratory direct and derived parameters not only allow assessment of overall cardiopulmonary reserves, but also in the case of a limitation of exercise capacity, that is, a reduced VO2max, indicate the reason for this, such as pulmonary, cardiovascular, or muscle-skeletal limitations. If a particular organ system can be incriminated as the limiting factor, specific treatment options can be of benefit, such as optimization of COPD treatment or management of ischemic heart disease.
A recently published meta-analysis including 955 patients from 14 studies confirmed that the VO2peak was lower in patients developing postoperative complications expressed either as absolute value or percentage of predicted, supporting the currently proposed thresholds that are based on individual studies with smaller number of patients.[27]
Likewise, a recent article showed an increased risk of complications and mortality after lung resection when the preoperative VO2peak was less than 15 ml/kg/min or 65% of predicted.[15]
In a consecutive series of more than 100 patients with a VO2peak measured during cycle ergospirometry less than 15 ml/kg/min and submitted to lung resection, we found morbidity and mortality rates of 25 and 3.9%, respectively. All patients who died had a VO2peak value of less than 12 ml/kg/min and were submitted to major lung resection (mortality rate of 15% in the group with VO2peak <12 ml/kg/min) (Brunelli A, personal communication).
Residual Function and Quality of Life
What price is the patient ready to pay for radical resection? For many patients, the risk of an impaired QOL and residual function after surgery is an important consideration when deciding whether to proceed with surgery. Some of them may be ready to accept the risk of immediate cardiopulmonary complications but are not prepared to accept significant postoperative functional debility.[28]
It has been recently demonstrated that 1 month after lobectomy, FEV1, DLCO and VO2peak values were 79.5, 81.5 and 96% of the preoperative values, respectively, and recovered up to 84, 88.5 and 97% after 3 months, respectively. On the contrary, 1 month after pneumonectomy FEV1, DLCO and VO2peak values were 65, 75 and 87% of the preoperative values, respectively, and increased to 66, 80 and 89% after 3 months, respectively.[4] Although other authors[29] have found slightly greater reductions in FEV1 and VO2peak at 6 months, it appears that after lung resection, patients had a more complete recovery of their exercise tolerance compared with their airflow and gas-exchange capacities, presumably due to other compensatory mechanisms related to the cardiovascular system and the peripheral oxygen extraction capacity.
In line with previous reports,[30] we found that the exercise capacity after major lung resection was poorly correlated with FEV1 or DLCO values, indicating its multifactorial nature and the limited role of pulmonary function tests (PFTs) alone in predicting the residual postoperative physical activity.
We also found that 27 and 34% of patients with moderate to severe COPD improved their FEV1 and DLCO, respectively, at 3 months compared with their preoperative values.[9]
Most of the lung cancer patients are psychologically depressed and exhibit an increased tension-anxiety status compared with the general population before being submitted to operation.[31-33] Brunelli et al.[32] found that compared with the general population, candidates for lung resection with lung cancer had reduced preoperative values of role limitation caused by physical problems, general health perception, social functioning, role limitation caused by emotional problems, and mental health perception scales. All these scales, with the exception of general health, remained below 50 (general population mean) even 3 months after operation.
From the most recent evidences, it appears that lung resection determines a transient worsening of QOL at 1 month following the operation with most of the scales returning to preoperative values at 3 months.[32,34] Exception to this trend is represented by pneumonectomy patients that display a persistent deterioration of physiological and mental QOL scales even at 3-6 months after operation.[32-35]
Interestingly, in patients traditionally deemed at increased surgical risk (those with cardiac disease, COPD, ppoFEV1 or ppoDLCO or all <40% or older than 70 years of age) the postoperative physiological or mental statuses did not differ from those of lower risk counterparts.[32] These findings may have great importance during patient counseling before the operation. Particularly in patients deemed at higher risk for postoperative major cardiopulmonary complications, the information that residual QOL will be similar to the one experienced by younger and fitter patients may help in their decision to proceed with surgery. This information will be of paramount importance even for the referring physicians, who will be less reluctant to refer high-risk lung cancer patients to surgery.
Another interesting finding is that traditional parameters used to assess the postoperative cardiorespiratory function do not seem to correlate well with the QOL reported by patients.[32] Thus, PFTs and exercise tests cannot be taken as surrogates for QOL evaluation, as discrepancies may exist between patients’ perception about their residual physical and emotional status and objective functional measures. An ad-hoc instrument should always be used for QOL evaluation.
Conclusion
Most recent evidence shows the relative role of ppoFEV1 in assessing the risk of lung-resection candidates. DLCO should be estimated systematically before lung resection for it predicts complications even in patients with normal FEV1. The use of low-technology exercise tests should be reconsidered. Poor performance at stair-climbing test is associated with increased morbidity and mortality. Given its cost-effectiveness this test seems ideal as a first-line screening test. However, VO2max should be directly measured by cycle ergospirometry in patients with an impaired performance at low-technology exercise tests. From the findings of this review, we propose an institutional functional algorithm in which the roles of DLCO and exercise tests are emphasized (Figure 1).
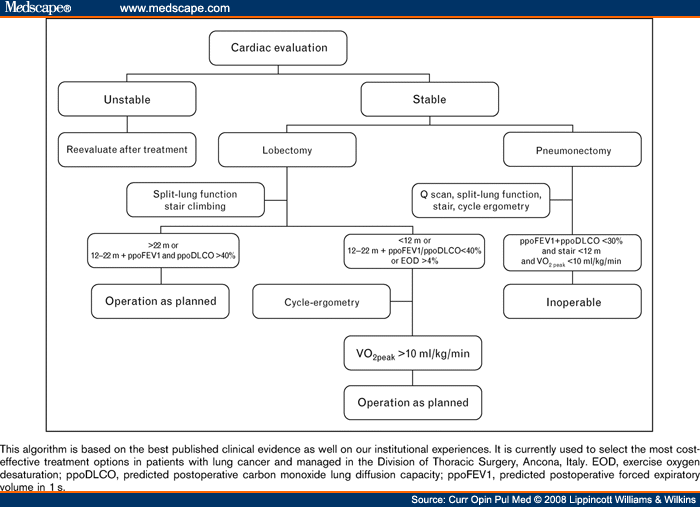
Figure 1.Functional algorithm for the evaluation of lung-resection candidates
References
|
Alessandro Brunelli, MD, Via S. Margherita 23, 60129 Ancona, Italy Tel: +39 0715964439; fax: +39 0715964433; E-mail: alexit_2000@yahoo.com .
Tags: polmone
